Underneath is a video of Myriam Hunink, MD, PhD explaining the DESTRESS study:
…
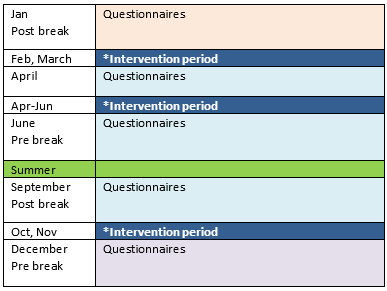
Every participant of the DESTRESS study is part of our cohort. You will receive questionnaires at specific time points, see the figure below. We want to study how the stress levels of students at the Erasmus MC change over time. Depending on your answers to the questionnaires, you may be eligible to follow a resilience training program. Please read the information on this website before subscribing.
Participants can be all students (Bachelor, Master, Doctoral, PhD) in fields related to health care, public health, medicine, psychology and life sicences at, or affiliated with, the Erasmus University Rotterdam.
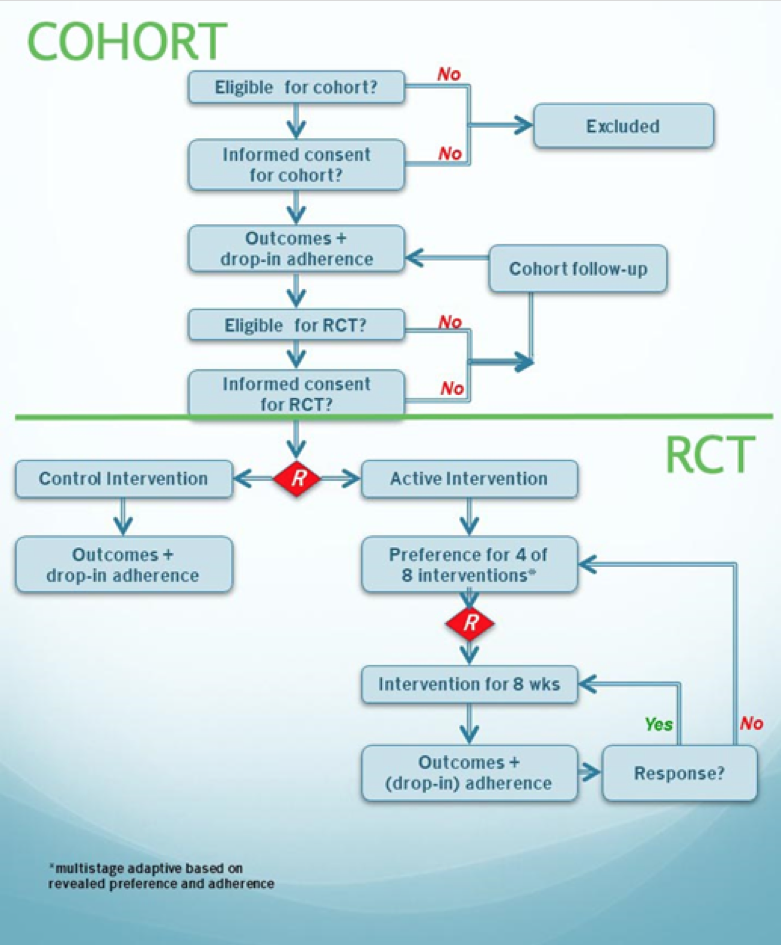
Joining the RCT part of the DESTRESS study is optional. Once you give informed consent and you are eligible, you will be randomized to the intervention or the control group. If you are randomized to the intervention group, you will be randomized to one of your preferences. The first round the randomization takes place over your top 4 preferences, second over your top 3 and third (and last) over your top 2.
FAQ
Can I participate in the study? To be eligible for participation, you have to be a student in a healthcare-related degree program. This includes students in medicine, biomedical research, health policy and management, health economics (and HEPL), psychology, nanobiology, clinical technology, and life sciences. You also have to be 16 years or older and have health insurance.
Furthermore, it is important that you have access to internet as the questionnaires will be distributed per email. It is also important that you are able to understand English as the questionnaires and information are all in English. Group-based interventions will be in English or Dutch or both, as appropriate for the participants in that group.
Even if you do not feel stressed, it is still important and useful that you participate in this study: both for yourself and for future students! Participation in the study provides you (and us) with information about chronic stress and the prevention of burnout. You will learn to recognize early symptoms of chronic stress and burnout and how to cope with stress. The programs are offered to our students free of charge. The programs are also very accessible: some are online, some are group-based programs and take place in or around the Erasmus MC after 5 pm.
Chronic stress and burnout can negatively influence your health and well-being, your study results, and your career. By teaching you stress management skills, we hope to help you to prepare yourself for the future. With your participation in the DESTRESS study, we hope to learn more about chronic stress and burnout among students. Your participation will not only be helpful to you: it will also help us develop programs for future students.
Each program has a duration of 8 or 9 weeks. If you do not like the program you have chosen or the program has not been beneficial for you, you have the option to switch to another program after the first round of 8 or 9 weeks.
It is important that you properly weigh the possible benefits and disadvantages before you decide to participate in the study.
Benefits
- Participation in the cohort part of the study will provide you with insights into your perceived stress and your well-being and will inform you about stress reduction and burnout prevention.
- Participation in the RCT part of the study will give you the opportunity to participate in programs that reduce chronic stress and prevent burnout
- The programs will increase your resilience, a skill that will be beneficial to you now during your studies and in your future career
- The programs are offered free of charge to you.
- The programs are held in or around the Erasmus MC, so it is easy to attend the programs.
- The duration of a program is 8 or 9 weeks. After these 8 or 9 weeks you can decide whether
- you want to stop with the program.
- you want to switch to another program of the study.
- you want to continue with the program.
- The programs consist of low-risk practices.
Disadvantages
- Participation in this study costs time. Filling in the questionnaires will take around 20-30 minutes. You have to fill in 7 online questionnaires over a period of 2 years. The group sessions of the programs will take 1.5-2 hours per week and participants are encouraged to do 30-45 minutes of exercises at home 5 days a week.
- Although there is evidence for benefit of the programs that we offer in reducing symptoms of chronic stress and preventing burnout, there is uncertainty about the benefits. The degree of benefit is uncertain and no guarantees can be given for a beneficial effect in everyone.
- Some of the programs involve physical activities. All offered programs are low risk activities. As with all exercise, there is always a small risk of injury. Make sure you inform the trainer of any physical limitations and injuries you have prior to embarking on the activities. Challenge yourself but do not overdo it!
Before you decide whether you want to participate in this study, it is essential that you read the information carefully about what the study entails. This information can be found on the website or you can read the general brochure on medical research. Do not hesitate to ask the investigator or the independent expert, who are mentioned on the page ‘Contact’, for additional information. You may also discuss it with your partner, friends or family.
Once you have consented to take part in the study, you will be asked to fill in a set of questionnaires. All questionnaire responses will be treated confidentially: there will be no connection made to your degree program or teachers. The set of questionnaires will be sent seven times over the course of the study: six times within the first year with about 2 months between every set of questionnaires. Two years after the initial start, you will be asked to fill in the last set of questionnaires. Filling in the set of questionnaires takes roughly 20-30 minutes. This is the cohort part of the study as shown in figure 1. (Figure 1 can be found at the end of this paragraph.)
Depending on your answers to the first set of questionnaires, you may be eligible to follow a resilience training program. For this part of the study, we will again ask your consent. You will be randomized to either care as usual (control) or to the resilience training program. There are different programs: Mindfulness-Based Stress Reduction (MBSR), running, yoga, aikido, music and stress management training. Some are online, some are group-based. We will ask you to indicate your preference for which interventions you would like to try in the questionnaire. For more information about the different interventions, we refer you to the page ‘Resilience training programs’. You will then be randomized to one of your preferred programs. This is the RCT part of the study and this is also shown in figure 1. If you take part in the RCT part of the study, you will be asked to fill in the same questionnaires as students participating in the cohort study.
The duration of a program is 8 or 9 weeks. If there is no beneficial effect of the followed program, you can choose to switch to another program and follow that program for another 8 or 9 weeks. If there is a beneficial effect of the followed program, we encourage you to continue with maintenance practice at home or in a group for continued benefit.
It is up to you to decide whether you take part or not. You can decide to participate in only the cohort part of the study or also participate in the RCT should you be selected. Even after you have given your permission to participate in the study, you are still free to withdraw consent from the study at any time and you do not have to give a reason for withdrawing.
Figure 1 shows the cohort part and the randomized control trial (RCT) part of the study. We will ask you to give separate consent for the cohort part and the RCT part of the study. R stands for randomization.
We ask you to fill in all of the questionnaires as completely and honestly as possible. It only takes 20-30 minutes to fill in one set of questionnaires. We will ask you to do this seven times over the course of two years.
If you are selected for a resilience program, we expect you to participate, do the exercises, and show up at group sessions if applicable. In extraordinary situations, like sickness, it is okay to miss a session. We understand that everyone has responsibilities and that something can always happen. We do ask you to contact the trainer of the program if you are not able to be present at the session. The contact details of the trainer can be found in the email that you will receive after you have been selected for a program. Depending on which program you will follow, the length of a session can vary from 1 to 2 hours. Be sure to check the page ‘Resilience training programs’ for the exact times, and for more information. Some additional information will be sent to your email account if you are selected for that particular program.
There are a few situations where it is important that you contact us by using the contact form:
- If you are admitted to hospital or are going for treatment there.
- If you suddenly develop any health problems.
- If you no longer want to participate in the study.
- If your contact details change.
The most important thing is to enjoy yourself during the sessions! The programs are offered to you free of charge and we hope that through the lessons, you will develop stress management skills that will help you now and in the future.
If you meet the criteria and you have read all of the information on this website, you can sign up for participation by clicking on the “register” button below. Registration is now closed. The resilience training programs will start in the week of April 19th.

Currently you will not receive ECTS for participation in the DESTRESS study. For more information please contact your study advisor or helpdesk.
The Resilience training programs are completely free of charge. Materials and equipment for the programs will be provided without additional costs.
You will not be paid for your participation in this study and you will not be reimbursed for your travel costs.
It is up to you to decide whether or not to participate in the study. Participation is voluntary.
If you do participate in the study and you change your mind, you can withdraw at any time during the study. You do not have to give a reason for withdrawing. The data collected until that time will still be used for the study. If you want to stop participating, please send a mail through to contact form with your email that is registered in this study. We will then ask your permission for using your collected data and for keeping your data for 15 years after study closure.
Your participation in the study stops when one of the following applies:
- you have completed all questionnaires.
- you choose to stop.
- the end of the entire study has been reached.
- the independent psychologist considers it is best for you to stop.
- the Erasmus Medical Centre Rotterdam, the Data and Safety Monitoring Board or the Medical Research Ethics Committee, decides to stop the study.
The study is concluded once all the participants have completed the study. After processing the data, the investigator will provide the most important results of the study on the website.
Your personal data will be collected, used and stored for this study on a protected server. This concerns data such as your name, date of birth, email address, and data about your health. The collection, use and storage of your data is required in order to answer the research questions of the study.
After removal of personal identifying information, the data will be analyzed in the context of the cohort study, the RCT, and a study with pooled data of students in other cities and countries in similar studies. We ask your permission for the use of your de-identified data for these analyses.
Confidentiality of your data
To protect your privacy, your data will be given a code. Your name and other information that can directly identify you, will be omitted. Data can only be traced back to you with the encryption key. The encryption key remains safely stored in the local research institute. The data that is sent to the sponsor will only contain the code, not your name or other data with which you can be identified. The data cannot be traced back to you in reports and publications about the study. Your data will not be given to university staff, it will not be given to teachers or professors, and we do not ask the university for any information about you.
Access to your data for verification
Research assistants have access to part of your data for study logistics: they enter your email address into the system and safeguard the sending of the correct emails. Some people can access all your data at the research location, including the data without a code. This is necessary to check whether the study is being conducted in a reliable and safe manner and according to good clinical practice. Persons who have access to your data for review are the committee that monitors the safety of the study, a monitor and auditor working for the Erasmus MC, and an auditor working for the national authorities. They will keep your data confidential. The handling of personal data will comply with the Dutch Personal Data Protection Act (in Dutch: de Wet Bescherming Persoonsgegevens). We ask you to consent to this access.
Storage and use of data for other research
Your data may also be of importance for other scientific research in the field of chronic stress and burnout. To this end, your data will be kept for 15 years after study closure as stated in the Dutch law (article 454 of the Burgerlijke Wetboek).
Withdrawing consent
You can withdraw your consent to the use of your personal data at any time. This applies to this study and also to storage and use for future research. The study data collected until the moment you withdraw your consent will still be used in the study.
More information about your rights when processing data
For general information about your rights when processing your personal data, you can consult the website of the Dutch Data Protection Authority.
If you have questions about your rights, please contact the person responsible for the processing of your personal data. Contact details can be found on the page “Contact”.
If you have questions or complaints about the processing of your personal data, we advise you to first contact the research location. You can also contact the Data Protection Officer of the institution or the Dutch Data Protection Authority. You can find the contact details on the page “Contact”.